Separating Solids
Grade Level: Grade 9
Discipline: Integrated Chemistry and Physics, Filtration, Physical Properties
How do you separate a mixture that contains two solids/substances when one of them is soluble (salt) and one of them is insoluble (sand)?
For the first component of this lab, students receive a bag filled with pebbles, sand, chalk, and iron filings. Students then use a magnet and sieve cups to separate the different parts of the mixture (see first 2 pages of attached lab).
For the second component of this lab, students receive a mixture of sand and salt (they take the initial mass) and must separate the insoluble sand from the soluble salt with warm water. Students then check on their sand and salt cups each day until fully dry, where they take the masses again to find percent composition of the sand and the salt.
Learn more by downloading the lab above.
Recommended Products
[StartProductBlock]

Separation of a Mixture of Solids
|
One of the many methods used to separate a mixture is liquid-liquid extraction or solvent extraction. In this laboratory, a mixture of two compounds in a specific ratio will be separated using an aqueous/organic solvent system, and the percent yield will be determined in order to demonstrate the law of conservation of matter.
[EndProductBlock]
[StartProductBlock]
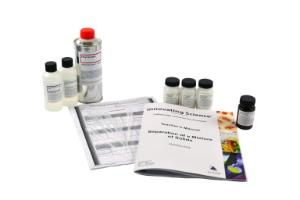
Separation of a Mixture of Solids - Small Group Learning Kit
Performing liquid-liquid extractions allows students to apply their knowledge of the properties of molecules. Students will learn how the properties of solubility can be utilized to separate two solids in a mixture.
[EndProductBlock]
[StartProductBlock]

Soil, Sands, and Gravel Set
Useful for your weathering and erosion, soil formation, porosity and permeability, or other general earth science labs.
[EndProductBlock]
